Before you can understand what monoclonal antibodies are, you need a good understanding of antibodies in general. I won’t go through everything here so read this article if you’re not already confident.
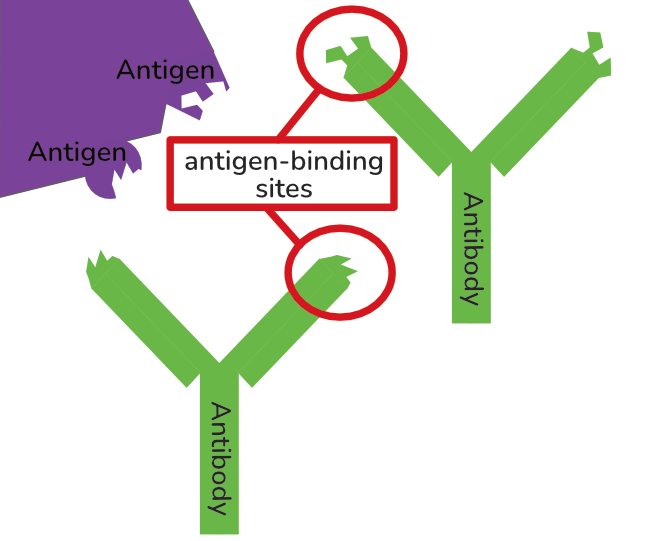
To summarise as a recap: antibodies are small protein molecules with variable antigen-binding sites. They bind molecules that don’t belong in the body to flag these up to the immune system. Eg they might bind to a viral surface protein, or a bacterial polysaccharide. The thing that they bind is called an “antigen”.
Monoclonal Antibodies
Mono = one (e.g. monomer, monosaccharide, monoxide)
Clone = an identical copy of a cell/organism with the same DNA, created from one original cell/organism (e.g. clonal selection; clonal expansion; Attack of the Clones)
Antibody = a protein molecule that binds antigens, mediating an immune response
Monoclonal antibodies are identical antibodies, made by B-lymphocytes cloned from one single starter cell.
Why inject Monoclonal Antibodies
Normally, antibodies are synthesised and released in the body by B-lymphocytes. But this requires two things: firstly that the immune system is aware of a threat, and secondly that there is a T-lymphocyte with DNA that encodes the required antibody.
The T-lymphocyte is necessary as it’s involved in sparking off B-lymphocyte replication and antibody production. But also the T-lymphocyte provides a check that it’s safe to use the antibody.
In my case, my body isn’t aware that it would be helpful to make antibodies to that pesky migraine-provoking protein. And I almost certainly don’t have any T-lymphocytes that would give the OK to produce such an antibody. At least, I shouldn’t do. Any such T-lymphocytes should have been destroyed early in my life, along with all other T-lymphocytes that were capable of producing antibodies against my own body. So I need to get the antibodies from somewhere else.
Designer Antigen-Binding Sites
In the lab, you can make any antibody you want. You just need the right B-lymphocyte.
There are a few different ways to tinker with the genetic code of a B-lymphocyte to achieve this. You don’t need to know the details. But what you do need to understand is that inside the B-Lymphoctyle, the scientist needs to ensure that the section of its DNA that codes for the antibody’s antigen-binding site has a sequence that …
… will be translated during protein synthesis into a chain of amino acids which ….
… contains a particular sequence of amino acids (primary structure) so that …
… the chain folds its backbone (secondary structure) in a way that allows …
… the whole thing to fold up upon itself (tertiary structure) so that it …
… presents a binding site with a specific shape and chemical properties that …
… will bind the antigen that they want it to bind.
This one cell can then be cloned. This produces many many identical, cloned cells with that exact same DNA, capable of producing identical antibodies with identical binding sites. Remember mono = one. This is where the “monoclonal” comes from.
Make big vats of these monoclonal cells and you can get them to pump out huge numbers of your chosen antibody to be collected and purified to use as you wish. These are monoclonal antibodies. Each antibody molecule is identical because the cells are all identical clones with the same DNA sequence.
The monoclonal antibodies in my injection pens were made like this in a lab. They have an antigen-binding site that is able to bind a protein called CGRP. By doing so, they prevent the CGRP from binding to its natural receptor, including in a particular set of neurons in my head. Which prevents my migraines.
But monoclonal antibodies can do a lot more than this - they are highly versitile due to their small size and specific binding …
Weaponising Antibodies as Therapeutics
Why stop just with changing the binding site?
Monoclonal antibodies specifically bind to your target, encumbering it and provoking a natural immune response. But why not go further? Why not get the antibody to deliver a powerful weapon directly to its target?
A big problem with injected/ingested drugs is that they get everywhere. If you inject a chemotherapy drug, it travels through the bloodstream without any map or guidance system. It reaches every part of the body. Cancer drugs usually target fast-dividing cells, but this means that as well as damaging the cancer, they get into your hair follicles where they kill healthy cells so that your hair falls out. They get into cells in your gut and kills them, making you feel sick and suffer gastrointestinal problems.
But what if you attached the drug to a monoclonal antibody that only binds the target cancer cells? It will still travel around the body in the blood, but will stop at the cancer and have much greater impact there.
Monoclonal antibodies are used in cancer therapies not only to provoke a normal immune response, but also to deliver cancer drugs, or stick cell-killing radioactive substances onto individual cancer cells. Being able to target the cancer in this way reduces unpleasant side-effects and so broadens the range of drugs that can be used.
Monoclonal Antibodies in Diagnostics
Monoclonal antibodies are useful tools outside the body too.
Until the 1950’s or so, pregnancy tests were carried out using live frogs. They would inject the woman’s urine, and if she was pregnant then her hormones would cause the frog to produce eggs just over a week later. Happily for frogs, we do things a bit differently now. (You don’t need to know about the frogs, although you may now never forget that mental image. You’re welcome.)
The modern pee-on-a-stick pregnancy test is a Lateral Flow Device. They work in very much the same way as Covid tests. You add body fluids, which soak their way along an absorbant strip, and if a certain molecule is present (eg a particular pregnancy hormone, or viral coat proteins) then a visible line appears. How do they detect the molecule of interest? By using monoclonal antibodies that will specifically bind to it. Similar tests can also be used to detect prostate cancer or HIV.
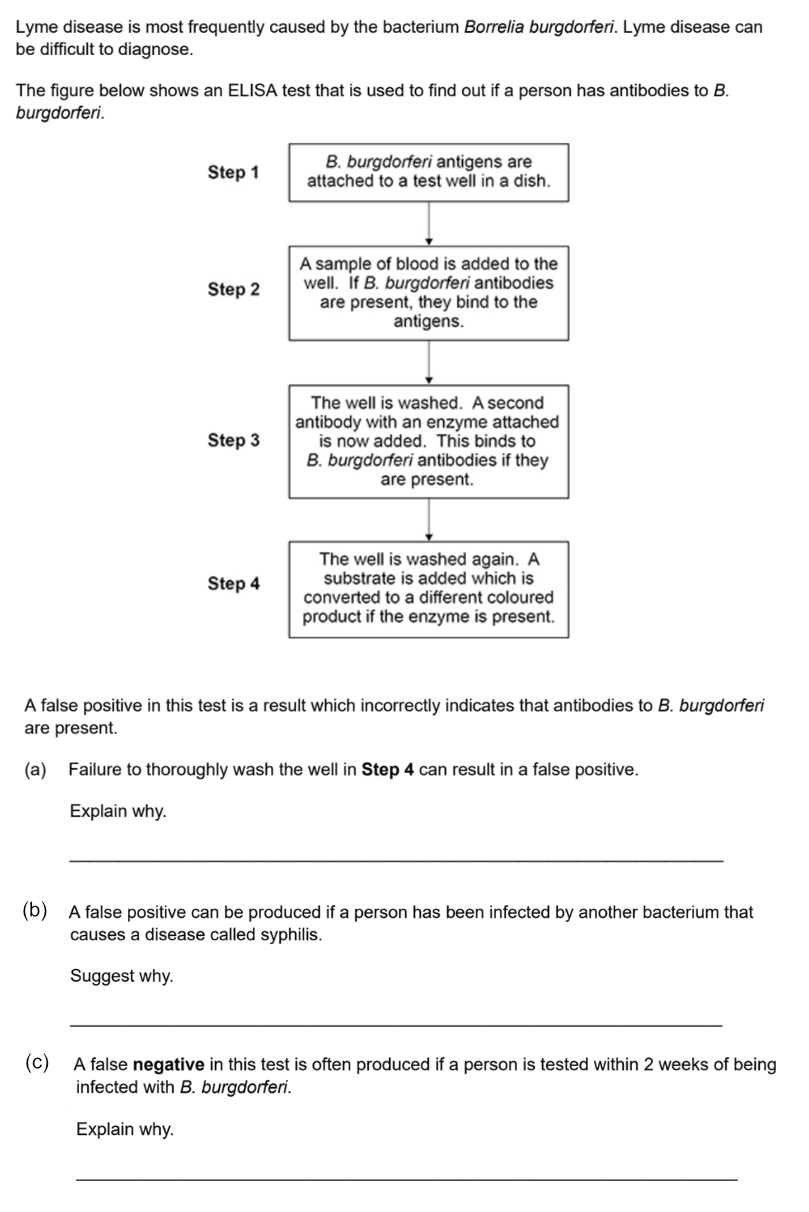
ELISA tests (for AQA)
ELISA tests work in a similar way, biochemically speaking. There are different versions but here’s the one it’s most important to know about. ELISA tests can be confusing because different types of antibodies play different roles in the process.
Direct ELISA test - a test to detect antibodies in the blood
If you are infected with a pathogen, your body will react by producing antibodies that are able to bind antigens associated with that pathogen. By detecting these antibodies, you can be diagnonised as being infected.
Here is how the test works, step by step:
1. An antigen from the pathogen (eg a viral coat protein) is covalently bonded to the well surface.
2. Blood plasma is put into the well. If antibodies for this antigen are present in the blood, they will bind to the antigen.
3. The blood plasma is washed out of the well, leaving behind any antibodies bound to the antigen.
If there are antibodies in the well, then you know the person has had an immune response to the pathogen. But how can you tell if antibodies are there or not? They’re such tiny proteins.
A totally different type of antibody is used for the next step. It’s a monoclonal antibody made in the lab, but it’s also a very unusual one. It is an unnatural, designed tool created purely for use in biochemical assays. These antibodies have some very special properties:
• Their antigen-binding sites specifically bind to the constant region of natural antibodies. This means that for these monoclonal antibodies, other antibodies are antigens! (Yes this is confusing, but it’s a good way to check you really understand what ‘antigen’ means.)
• Their constant region is covalently bonded to an enzyme. The presence of the enzyme means that they can’t bind each others’ constant regions - so they are not antigens to themselves. They only bind other types of antibody.
Imagine the chaos in your body if your B-cells released antibodies that could bind to other antibodies’ constant regions! They would be hugely damaging to your immune system. However, these little guys are very useful tools in the lab.
5. These special monoclonal antibodies, with linked enzyme, are added to the well.
• If there ARE (natural) antibodies bound to the antigen in the well, the monoclonal antibodies will bind to their constant region.
• If there are NO (natural) antibodies, the monoclonal antibodies will remain freely floating in the solvent.
6. The well is washed out again.
The monoclonal antibodies, with their linked enzyme, will only remain in the well IF there were (natural) antibodies in the blood sample. Otherwise they would have been washed away in step 6. If there is enzyme in the well, there must have been antibodies in the blood.
But how do we know if there is enzyme in the well..?! This bit is easy, because of the clever choice of enzyme: The enzyme is one that takes a colourless substrate to form a coloured product.
7. Add the substrate, and see what happens! If colour appears, you know the enzyme is present. And the enzyme if present, its monoclonal antibody must be bound to a natural antibody that could bind the antigen from the pathogen.
AQA Exam Question Example - ELISA tests
This exam question requires you to understand both ELISA tests and the immune response. Can you make sense of it?